临床功效
BB536改善胃肠道疾病
BB536可恢复最佳排便功能

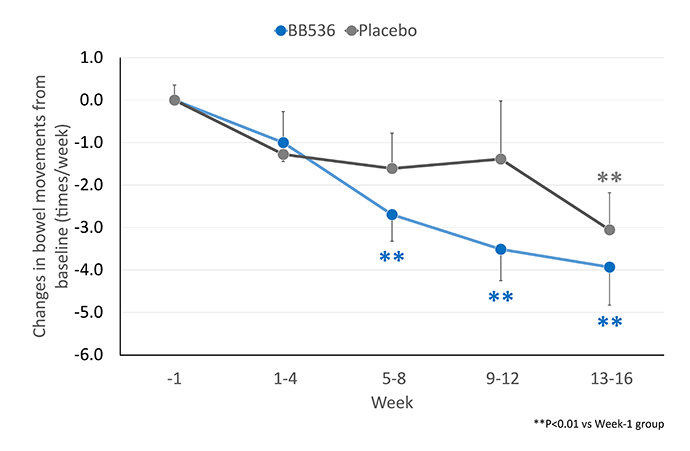
BB536长期以来被认为是改善胃肠道状况最有效的益生菌菌株之一。越来越多的临床证据表明,食用含有BB536的乳制品,包括酸奶、酸奶饮料和非发酵乳,可以改善便秘或腹泻的婴幼儿、儿童、成人和老年人的排便频率和粪便性状[1-4]。
在 168 名年龄 >65 岁接受肠内管饲的患者中[4],服用BB536(每日 250 亿和 500 亿CFU的低剂量和高剂量)16 周,与安慰剂对照组相比,排便次数少(≤4 次/周)的患者的排便情况得到了更好的改善。
服用BB536后,排便频率较高(≥10次/周)的患者也恢复了规律的排便。与安慰剂对照组相比,服用BB536的患者正常成形粪便的发生率显著更高。
Reference:
- 1Puccio, G., Cajozzo, C., Meli, F., Rochat, F., Grathwohl, D. and Steenhout, P., 2007. Clinical evaluation of a new starter formula for infants containing live Bifidobacterium longum BL999 and prebiotics. Nutrition, 23(1), pp.1-8.
- 2Russo, M., Giugliano, F.P., Quitadamo, P., Mancusi, V., Miele, E. and Staiano, A., 2017. Efficacy of a mixture of probiotic agents as complementary therapy for chronic functional constipation in childhood. Italian Journal of Pediatrics, 43(1), p.24.
- 3Wong, C.B., Odamaki, T. and Xiao, J.Z., 2019. Beneficial effects of Bifidobacterium longum subsp. longum BB536 on human health: Modulation of gut microbiome as the principal action. Journal of Functional Foods, 54, pp.506-519.
- 4Kondo et al., 2013. Modulatory effects of Bifidobacterium longum BB536 on defecation in elderly patients receiving enteral feeding. World Journal of Gastroenterology: WJG, 19(14), p.2162.
BB536改善肠道微环境
产肠毒素脆弱拟杆菌(ETBF)与结肠炎相关。
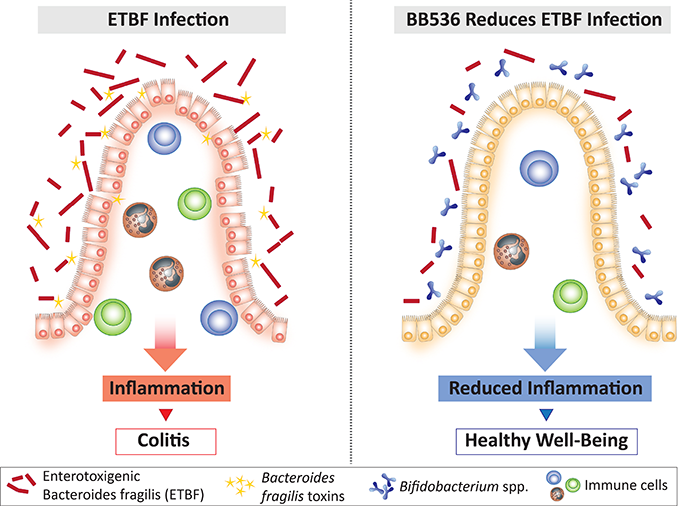
越来越多的证据表明,肠道菌群失调可能是胃肠道疾病发生的病因之一。特别是,已有报道指出,某些细菌,例如产毒素细菌——肠毒素脆弱拟杆菌(ETBF),与炎症性肠病(IBD)患者的急性和持续性腹泻[1]以及结直肠癌的发生[2,3]相关。
BB536消除了肠道中的ETBF
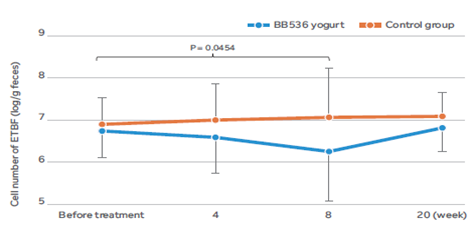
在 32 名携带 ETBF [4]的健康成年人中,连续 8 周摄入含有BB536的酸奶对肠道菌群中 ETBF 的细胞数量产生了明显的影响。
结果显示,与基线值相比,食用BB536酸奶的受试者肠道内ETBF细胞数量显著下降,而对照牛奶组则未观察到此现象。然而,当受试者停止食用BB536酸奶后,ETBF细胞数量恢复至基线水平。这些结果表明,持续摄入BB536可能清除肠道菌群中的机会性ETBF致病菌,并改善肠道微环境。
Reference:
- 1Sears, C.L., 2009. Enterotoxigenic Bacteroides fragilis: a rogue among symbiotes. Clinical Microbiology Reviews, 22(2), pp.349-369.
- 2Orberg, E.T., Fan, H., Tam, A.J., Dejea, C.M., Shields, C.D., Wu, S., Chung, L., Finard, B.B., Wu, X., Fathi, P. and Ganguly, S., 2017. The myeloid immune signature of enterotoxigenic Bacteroides fragilis-induced murine colon tumorigenesis. Mucosal Immunology, 10(2), p.421.
- 3Wu, S., Rhee, K.J., Albesiano, E., Rabizadeh, S., Wu, X., Yen, H.R., Huso, D.L., Brancati, F.L., Wick, E., McAllister, F. and Housseau, F., 2009. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nature Medicine, 15(9), p.1016.
- 4Odamaki, T., Sugahara, H., Yonezawa, S., Yaeshima, T., Iwatsuki, K., Tanabe, S., Tominaga, T., Togashi, H., Benno, Y. and Xiao, J.Z., 2012. Effect of the oral intake of yogurt containing Bifidobacterium longum BB536 on the cell numbers of enterotoxigenic Bacteroides fragilis in microbiota. Anaerobe, 18(1), pp.14-18.
BB536调节免疫反应
BB536可降低流感发病率,这可能是通过增强先天免疫力实现的。
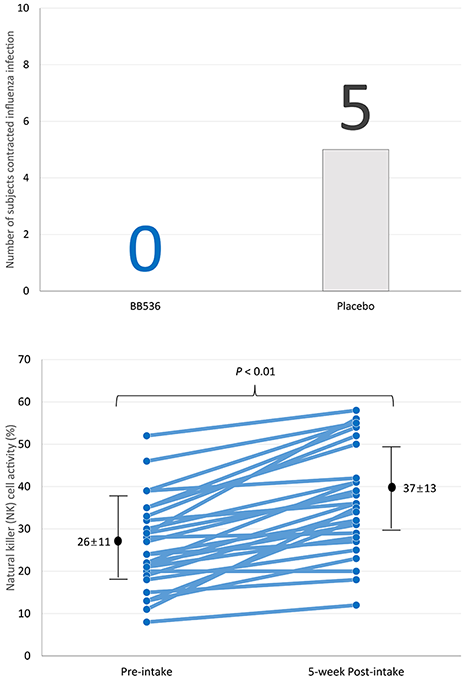
多项临床研究表明,BB536能够调节人体的免疫反应,尤其是在老年人群中[1]。据报道,老年人的细胞免疫反应较弱,免疫系统功能的下降被认为会导致疫苗效力降低,并增加流感病毒感染的风险。重要的是,BB536已被证实能够改善老年人逐渐减弱的免疫力[2,3]。
27名65岁及以上的老年人预先服用BB536粉剂(1000亿CFU /天),持续5周,并在第3周接种流感疫苗。随后,受试者被随机分为BB536组(n=13)和安慰剂组(n=14),持续14周。研究期间,服用BB536组的流感症状患者人数少于未服用组。此外,服用BB536后,自然杀伤细胞活性和中性粒细胞杀菌活性均有所增强,表明免疫力得到提升[3]。
这些结果表明,BB536可能增强老年消费者对致病病毒的抵抗力,并可作为潜在的佐剂,用于改善老年人对流感疫苗的免疫反应。
Reference:
- 1Wong, C.B., Odamaki, T. and Xiao, J.Z., 2019. Beneficial effects of Bifidobacterium longum subsp. longum BB536 on human health: Modulation of gut microbiome as the principal action. Journal of Functional Foods, 54, pp.506-519.
- 2Akatsu, H., Iwabuchi, N., Xiao, J.Z., Matsuyama, Z., Kurihara, R., Okuda, K., Yamamoto, T. and Maruyama, M., 2013. Clinical effects of probiotic Bifidobacterium longum BB536 on immune function and intestinal microbiota in elderly patients receiving enteral tube feeding. Journal of Parenteral and Enteral Nutrition, 37(5), pp.631-640.
- 3Namba, K., Hatano, M., Yaeshima, T., Takase, M. and Suzuki, K., 2010. Effects of Bifidobacterium longum BB536 administration on influenza infection, influenza vaccine antibody titer, and cell-mediated immunity in the elderly. Bioscience, Biotechnology, and Biochemistry, 74(5), pp.939-945.
BB536缓解过敏性疾病
什么是过敏性鼻炎?
过敏性鼻炎,又称花粉症,是一种鼻腔炎症,通常由环境过敏原(如花粉、宠物毛发、灰尘或霉菌)引发。其症状包括流鼻涕或鼻塞、打喷嚏、眼睛发红、发痒、流泪以及眼周肿胀。例如,由日本柳杉花粉引起的季节性过敏性鼻炎是日本最常见的过敏性疾病之一,被认为是“国民病” [1]。日本柳杉花粉症(JCPsis)是一种由接触刺激性柳杉花粉引发的I型免疫球蛋白E(IgE)介导的过敏反应。
服用BB536可预防过敏性鼻炎
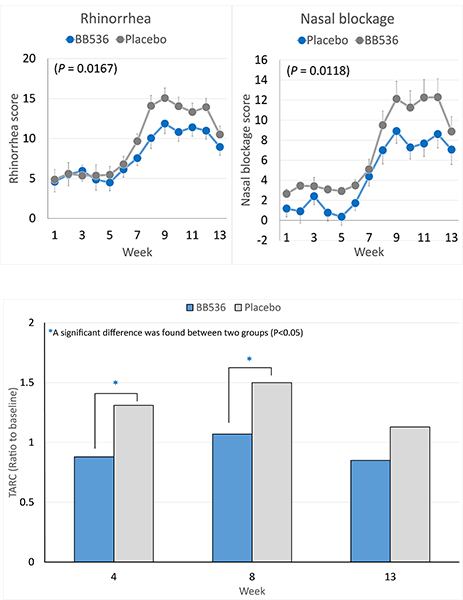
研究表明,摄入BB536酸奶或冻干粉可以缓解对日本柳杉花粉敏感的人的鼻部和眼部过敏症状,并调节过敏免疫反应[2-5]。
在 2005 年花粉最严重的季节,44 名 JCPsis 受试者(年龄 26-57 岁)[3]连续 13 周服用BB536粉末(500 亿CFU /2 克,每日两次)显著降低了流涕和鼻塞的过敏评分。
服用BB536显著改善了花粉传播引起的T辅助细胞2(Th2)偏向性免疫反应。与安慰剂组相比,服用BB536粉末的受试者血浆中胸腺和活化调节趋化因子(TARC)的水平显著恢复正常。
Reference:
- 1Yamada, T., Saito, H. and Fujieda, S., 2014. Present state of Japanese cedar pollinosis: the national affliction. Journal of Allergy and Clinical Immunology, 133(3), pp.632-639.
- 2Xiao, J.Z., 2006. Effect of Probiotic Bifidobacterium longum BBS36 in relieving clinical symptoms and modulating plasma cytokine levels in japanase cedar pollinosis during the pollen season. A randomized double-blind, placebo-controlled trial (vol 16, pg 86, 2006). Journal of Investigational Allergology and Clinical Immunology, 16(4), pp.273-273.
- 3Xiao, J.Z., Kondo, S., Yanagisawa, N., Takahashi, N., Odamaki, T., Iwabuchi, N., Miyaji, K., Iwatsuki, K., Togashi, H., Enomoto, K. and Enomoto, T., 2006. Probiotics in the treatment of Japanese cedar pollinosis: a double-blind placebo-controlled trial. Clinical & Experimental Allergy, 36(11), pp.1425-1435.
- 4Xiao, J.Z., Kondo, S., Takahashi, N., Odamaki, T., Iwabuchi, N., Miyaji, K., Iwatsuki, K. and Enomoto, T., 2007. Changes in plasma TARC levels during Japanese cedar pollen season and relationships with symptom development. International Archives of Allergy and Immunology, 144(2), pp.123-127.
- 5Xiao, J.Z., Kondo, S., Yanagisawa, N., Miyaji, K., Enomoto, K., Sakoda, T., Iwatsuki, K. and Enomoto, T., 2007. Clinical efficacy of probiotic Bifidobacterium longum for the treatment of symptoms of Japanese cedar pollen allergy in subjects evaluated in an environmental exposure unit. Allergology international, 56(1), pp.67-75.
