臨床研究
BB536改善胃腸道健康狀況
BB536可恢復最佳排便規律

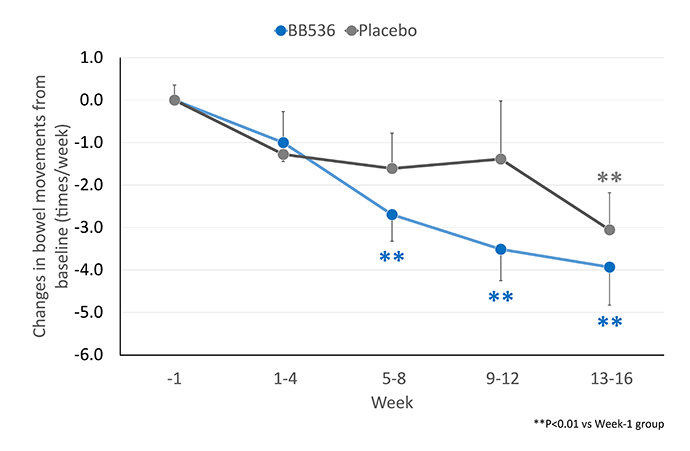
BB536長期以來被認為是改善胃腸道狀況最有效的益生菌菌株之一。越來越多的臨床證據表明,食用含有BB536的乳製品,包括優格、優格飲料和非發酵乳,可以改善便秘或腹瀉的嬰幼兒、兒童、成人和老年人的排便頻率和糞便性狀[1-4]。
在 168 名年齡 >65 歲接受腸內管餵食的患者中[4],服用BB536(每日 250 億和 500 億CFU的低劑量和高劑量)16 週,與安慰劑對照組相比,排便次數少(≤4 次/週)的患者的排便情況得到了更好的改善。
服用BB536後,排便頻率較高(≥10次/週)的患者也恢復了規律的排便。此外,相較於安慰劑組,攝取 BB536 可顯著提高正常成形糞便的比例。
Reference:
- 1Puccio, G., Cajozzo, C., Meli, F., Rochat, F., Grathwohl, D. and Steenhout, P., 2007. Clinical evaluation of a new starter formula for infants containing live Bifidobacterium longum BL999 and prebiotics. Nutrition, 23(1), pp.1-8.
- 2Russo, M., Giugliano, F.P., Quitadamo, P., Mancusi, V., Miele, E. and Staiano, A., 2017. Efficacy of a mixture of probiotic agents as complementary therapy for chronic functional constipation in childhood. Italian Journal of Pediatrics, 43(1), p.24.
- 3Wong, C.B., Odamaki, T. and Xiao, J.Z., 2019. Beneficial effects of Bifidobacterium longum subsp. longum BB536 on human health: Modulation of gut microbiome as the principal action. Journal of Functional Foods, 54, pp.506-519.
- 4Kondo et al., 2013. Modulatory effects of Bifidobacterium longum BB536 on defecation in elderly patients receiving enteral feeding. World Journal of Gastroenterology: WJG, 19(14), p.2162.
BB536改善腸道微環境
產腸毒性毒素脆弱擬桿菌(ETBF)與結腸炎有關。
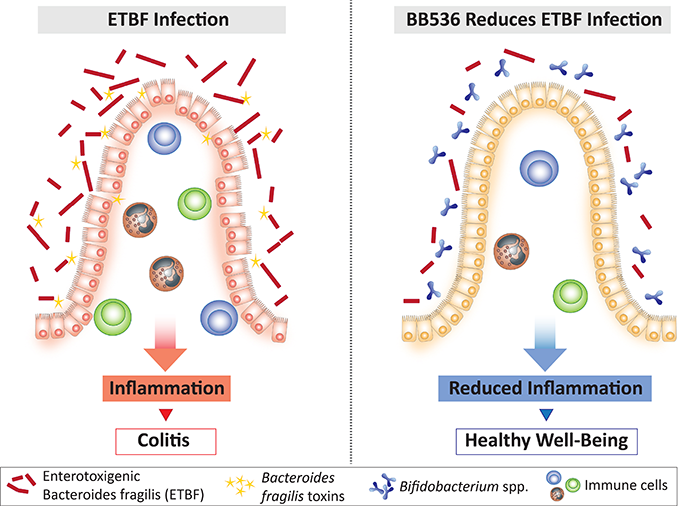
越來越多的證據表明,腸道菌叢失調可能是胃腸道疾病發生的原因之一。特別是,已有報告指出,某些細菌,例如產毒素細菌-腸毒素脆弱擬桿菌(ETBF),與發炎性腸道疾病(IBD)患者的急性和持續性腹瀉[1]以及大腸直腸癌的發生[2,3]相關。
BB536消除了腸道中的ETBF
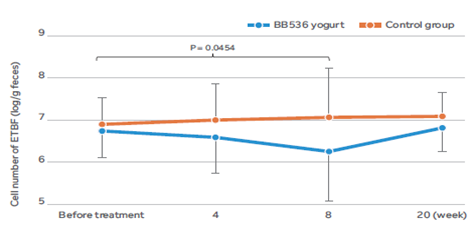
在 32 名攜帶 ETBF [4]的健康成年人中,連續 8 週攝取含有BB536的優格對腸道菌群中 ETBF 的細胞數量產生了明顯的影響。
結果顯示,與基線值相比,食用BB536優格的受試者腸道內ETBF細胞數量顯著下降,而對照牛奶組則未觀察到此現象。然而,當受試者停止食用BB536優格後,ETBF細胞數量恢復至基線。這些結果表明,持續攝取BB536可能清除腸道菌群中的機會性ETBF致病菌,並改善腸道微環境。
Reference:
- 1Sears, C.L., 2009. Enterotoxigenic Bacteroides fragilis: a rogue among symbiotes. Clinical Microbiology Reviews, 22(2), pp.349-369.
- 2Orberg, E.T., Fan, H., Tam, A.J., Dejea, C.M., Shields, C.D., Wu, S., Chung, L., Finard, B.B., Wu, X., Fathi, P. and Ganguly, S., 2017. The myeloid immune signature of enterotoxigenic Bacteroides fragilis-induced murine colon tumorigenesis. Mucosal Immunology, 10(2), p.421.
- 3Wu, S., Rhee, K.J., Albesiano, E., Rabizadeh, S., Wu, X., Yen, H.R., Huso, D.L., Brancati, F.L., Wick, E., McAllister, F. and Housseau, F., 2009. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nature Medicine, 15(9), p.1016.
- 4Odamaki, T., Sugahara, H., Yonezawa, S., Yaeshima, T., Iwatsuki, K., Tanabe, S., Tominaga, T., Togashi, H., Benno, Y. and Xiao, J.Z., 2012. Effect of the oral intake of yogurt containing Bifidobacterium longum BB536 on the cell numbers of enterotoxigenic Bacteroides fragilis in microbiota. Anaerobe, 18(1), pp.14-18.
BB536調節免疫反應
BB536可降低流感發生率,這可能是透過增強先天免疫力來實現的。
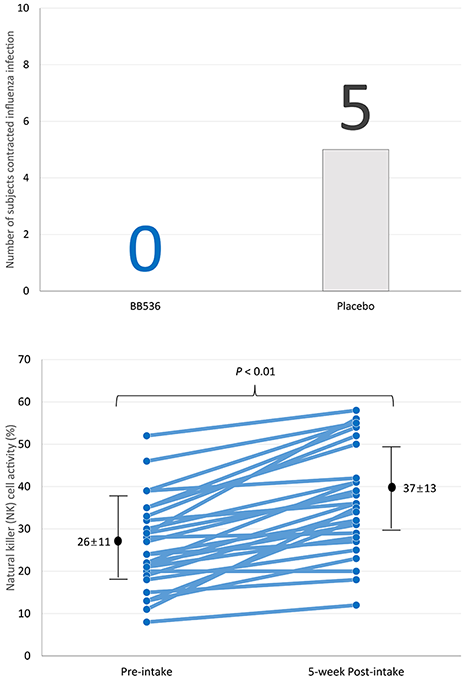
多項臨床研究表明,BB536能夠調節人體的免疫反應,尤其是在老年人群中[1]。據報道,老年人的細胞免疫反應較弱,免疫系統功能的下降被認為會導致疫苗效力降低,並增加流感病毒感染的風險。重要的是,BB536已被證實能夠改善老年人逐漸減弱的免疫力[2,3]。
27名65歲以上的老年人預先服用BB536粉劑(1000億CFU /天),持續5週,並在第3週接種流感疫苗。隨後,受試者隨機分為BB536組(n=13)和安慰劑組(n=14),持續14週。研究期間,服用BB536組的流感症狀患者人數少於未服用組。此外,服用BB536後,自然殺手細胞活性和嗜中性球殺菌活性均增強,顯示免疫力提升[3]。
這些結果表明,BB536可能增強老年消費者對致病病毒的抵抗力,並可作為潛在的佐劑,用於改善老年人對流感疫苗的免疫反應。
Reference:
- 1Wong, C.B., Odamaki, T. and Xiao, J.Z., 2019. Beneficial effects of Bifidobacterium longum subsp. longum BB536 on human health: Modulation of gut microbiome as the principal action. Journal of Functional Foods, 54, pp.506-519.
- 2Akatsu, H., Iwabuchi, N., Xiao, J.Z., Matsuyama, Z., Kurihara, R., Okuda, K., Yamamoto, T. and Maruyama, M., 2013. Clinical effects of probiotic Bifidobacterium longum BB536 on immune function and intestinal microbiota in elderly patients receiving enteral tube feeding. Journal of Parenteral and Enteral Nutrition, 37(5), pp.631-640.
- 3Namba, K., Hatano, M., Yaeshima, T., Takase, M. and Suzuki, K., 2010. Effects of Bifidobacterium longum BB536 administration on influenza infection, influenza vaccine antibody titer, and cell-mediated immunity in the elderly. Bioscience, Biotechnology, and Biochemistry, 74(5), pp.939-945.
BB536緩解過敏性疾病
什麼是過敏性鼻炎?
過敏性鼻炎,又稱花粉症,是一種鼻腔炎症,通常由環境過敏原(如花粉、寵物毛髮、灰塵或黴菌)引發。其症狀包括流鼻水或鼻塞、打噴嚏、眼睛發紅、發癢、流淚、眼周腫脹。例如,由日本柳杉花粉引起的季節性過敏性鼻炎是日本最常見的過敏性疾病之一,被認為是「國民病」[1]。日本柳杉花粉症(JCPsis)是一種由接觸刺激性柳杉花粉引發的I型免疫球蛋白E(IgE)介導的過敏反應。
服用BB536可預防過敏性鼻炎
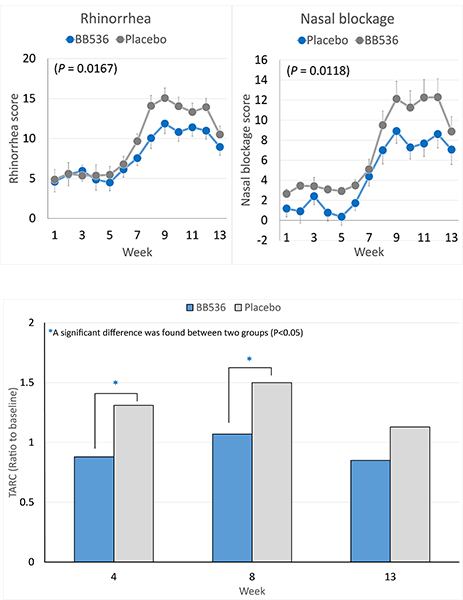
研究表明,攝取BB536優格或冷凍乾燥粉可以緩解對日本柳杉花粉敏感的人的鼻腔和眼部過敏症狀,並調節過敏免疫反應[2-5]。
在 2005 年花粉最嚴重的季節,44 名 JCPsis 受試者(年齡 26-57 歲)[3]連續 13 週服用BB536粉末(500 億CFU /2 克,每日兩次)顯著降低了流涕和鼻塞的過敏評分。
服用BB536顯著改善了花粉傳播引起的T輔助細胞2(Th2)偏向性免疫反應。與安慰劑組相比,服用BB536粉末的受試者血漿中胸腺和活化調節趨化因子(TARC)的水平顯著恢復正常。
Reference:
- 1Yamada, T., Saito, H. and Fujieda, S., 2014. Present state of Japanese cedar pollinosis: the national affliction. Journal of Allergy and Clinical Immunology, 133(3), pp.632-639.
- 2Xiao, J.Z., 2006. Effect of Probiotic Bifidobacterium longum BBS36 in relieving clinical symptoms and modulating plasma cytokine levels in japanase cedar pollinosis during the pollen season. A randomized double-blind, placebo-controlled trial (vol 16, pg 86, 2006). Journal of Investigational Allergology and Clinical Immunology, 16(4), pp.273-273.
- 3Xiao, J.Z., Kondo, S., Yanagisawa, N., Takahashi, N., Odamaki, T., Iwabuchi, N., Miyaji, K., Iwatsuki, K., Togashi, H., Enomoto, K. and Enomoto, T., 2006. Probiotics in the treatment of Japanese cedar pollinosis: a double-blind placebo-controlled trial. Clinical & Experimental Allergy, 36(11), pp.1425-1435.
- 4Xiao, J.Z., Kondo, S., Takahashi, N., Odamaki, T., Iwabuchi, N., Miyaji, K., Iwatsuki, K. and Enomoto, T., 2007. Changes in plasma TARC levels during Japanese cedar pollen season and relationships with symptom development. International Archives of Allergy and Immunology, 144(2), pp.123-127.
- 5Xiao, J.Z., Kondo, S., Yanagisawa, N., Miyaji, K., Enomoto, K., Sakoda, T., Iwatsuki, K. and Enomoto, T., 2007. Clinical efficacy of probiotic Bifidobacterium longum for the treatment of symptoms of Japanese cedar pollen allergy in subjects evaluated in an environmental exposure unit. Allergology international, 56(1), pp.67-75.
